 Treating Depression with Magnestism
Transcranial magnetic stimulation (TMS) is an experimental procedure that uses magnetic fields to alter brain activity.
There are different ways to perform the procedure. But in general, a large electromagnetic coil is held against your scalp near your forehead, often on the left side. An electric current creates a magnetic pulse, or field, that travels through your skull. The magnetic pulse causes small electrical currents in your brain. Those currents stimulate nerve cells in the region of your brain involved in mood regulation and depression.
Transcranial magnetic stimulation doesn't involve surgery, and you require no anesthesia. You also don't need to be hospitalized. Transcranial magnetic stimulation can be done on an outpatient basis in a doctor's office or clinic that's running a clinical trial for the procedure.
How transcranial magnetic stimulation works remains unclear
It's not clear precisely how transcranial magnetic stimulation may help relieve symptoms of depression. Networks of brain regions are thought to play a role in mood regulation. Stimulating the brain can change how it functions.
In some types of transcranial magnetic stimulation, brain activity is suppressed. In other types, brain activity is increased. In either case, the changes may be associated with improved mood. These improvements in symptoms may last for days or weeks.
In transcranial magnetic stimulation, a magnetic coil is held against your scalp, creating magnetic fields that alter brain chemistry and possibly boost mood.
Who may benefit from transcranial magnetic stimulation
Because it's experimental, transcranial magnetic stimulation isn't recommended as a first treatment for depression. It's used only as a research tool and is available in the United States only through clinical trials. In countries where transcranial magnetic stimulation has been formally approved to treat depression, it's typically used only for people with depression that hasn't improved with standard treatments or for those who may be considering electroconvulsive therapy but want an alternative.
People who have long-standing depression or depression that hasn't improved with standard treatments may be candidates for this experimental procedure. Talk to your doctor to see if it may be a good option for you.
Who should avoid transcranial magnetic stimulation
Certain people shouldn't have transcranial magnetic stimulation because of increased or unknown risks to their health. These include people:
Although transcranial magnetic stimulation is noninvasive, it still poses a risk of adverse side effects.
Of major concern is the possibility that transcranial magnetic stimulation can cause a seizure. Because of that risk, the International Society for Transcranial Stimulation advises that the procedure be performed only when medical help is quickly available and that those who administer it be trained as first responders who can provide emergency medical help.
Common side effects and adverse health problems associated with transcranial magnetic stimulation include:
Balancing the pros and cons of transcranial magnetic stimulation
For people with depression that doesn't get better with standard treatments, life can be miserable. Transcranial magnetic stimulation may provide hope that symptoms can improve. And for some people, it offers advantages over electroconvulsive therapy (ECT). Unlike ECT, transcranial magnetic stimulation doesn't typically induce convulsions, doesn't require anesthesia and may have fewer troubling side effects, such as memory loss.
However, research data about the effectiveness of transcranial magnetic stimulation are mixed. Some studies have shown that some people who undergo transcranial magnetic stimulation see a significant improvement in their symptoms that lasts for two weeks or more. Other research, though, has suggested that transcranial magnetic stimulation is no more effective than a placebo.
Researchers will learn more and perhaps hone the technique as more studies are done. Stronger stimulation done over a minimum of several weeks may be found more effective, for instance.
Stimulating The Brain Makes The Fingers More Sensitive
As reported in ScienceDaily, repetitive transcranial magnetic stimulation (rTMS) has emerged as an intriguing technique for exploring brain function, and a promising, though still unproven, form of therapy. This week, in the open-access journal PLoS Biology, Hubert Dinse and colleagues show that a short course of rTMS can increase finger sensitivity for up to two hours after treatment ends, and that this change corresponds to an increase in the size of the brain map representing the finger.
They tested the sensitivity of the index finger by determining how far apart two simultaneously applied pinpricks needed to be for the subject to distinguish them as separate stimuli. rTMS increased this two-point discrimination by about 15% immediately after stimulation, an effect that gradually diminished but still remained significant over the course of the next two hours. The effect was fairly specific for the right index finger: there was no effect on the left index finger, which is represented in the opposite hemisphere, and only a small effect on the right ring finger, which is represented several millimeters away from the index finger in the same hemisphere. When stimulation was applied over the area representing the lower leg, the index finger did not become more sensitive.
The authors used functional magnetic resonance imaging (fMRI) to see how the brain changed in response to the stimulation. They found that the region representing the index finger got larger, and that the degree of increase in any one subject corresponded to the degree of increased sensitivity in that same subject. As the sensory effect faded, so too did the fMRI changes. Thus, the cortex itself undergoes changes as a result of rTMS.
Notes:
1. Tegenthoff M, Ragert P, Pleger B, Schwenkreis P, Forster AF, et al. (2005) Improvement of tactile discrimination performance and enlargement of cortical somatosensory maps after 5 Hz rTMS. PLoS Biol 3(11): e362.
|
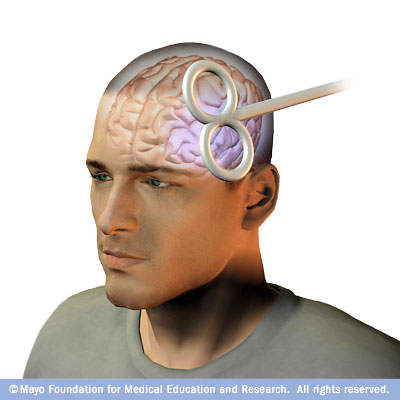 During transcranial magnetic stimulation, the magnetic coil is held against your head while electric current is switched on and off. You may feel a slight tapping or knocking sensation on your head. Although transcranial magnetic stimulation is generally painless, it may cause the muscles of your scalp or jaw to contract. The procedure lasts about a half-hour.
During transcranial magnetic stimulation, the magnetic coil is held against your head while electric current is switched on and off. You may feel a slight tapping or knocking sensation on your head. Although transcranial magnetic stimulation is generally painless, it may cause the muscles of your scalp or jaw to contract. The procedure lasts about a half-hour.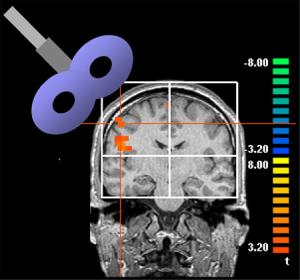 rTMS is applied with an electromagnetic coil in the shape of a figure-eight, placed on the scalp directly over the targeted portion of the brain. Short bursts of a strong magnetic pulse stimulate electrical currents within. Sensory input from each region of the body is represented on the surface of the brain, and the location of any region--in this case, the right index finger--can be mapped to allow precise targeting of the rTMS. The authors adjusted the strength of the magnetic field to just below that which triggered a sensory response in the finger, and then applied intermittent pulses of stimulation over the course of about ten minutes.
rTMS is applied with an electromagnetic coil in the shape of a figure-eight, placed on the scalp directly over the targeted portion of the brain. Short bursts of a strong magnetic pulse stimulate electrical currents within. Sensory input from each region of the body is represented on the surface of the brain, and the location of any region--in this case, the right index finger--can be mapped to allow precise targeting of the rTMS. The authors adjusted the strength of the magnetic field to just below that which triggered a sensory response in the finger, and then applied intermittent pulses of stimulation over the course of about ten minutes.