

Scientists Discover Chemical That 'Cancer-proofs' Naked Mole Rat's Cells
As reported in ScienceDaily, two researchers at the University of Rochester have discovered the chemical that makes naked mole rats cancer-proof. This is big news -- for the rats and for humans!
The findings could eventually lead to new cancer treatments in people, according to the study authors Andrei Seluanov and Vera Gorbunova. Their research paper will be published in the June 2013 edition of the journal, Nature.
The Backstory: Understanding what cancer actually is...
In a previous viewzone article I wrote about why we get old, worn out and eventually die. It has to do with a string of molecules that grows on each end of our DNA molecule. Called a "telomere" this substance has no apparent reason for existing except to provide a kind of blank leader -- like the blank film on an old reel movie -- that protects the vital DNA code each time it makes a copy of itself.
If we could only prevent the telomere from wearing down and shortening -- exposing the vital DNA to errors of replication -- then we could be "immortal!"
The amount of times the DNA can reproduce before this vital telomere is worn away is called the Hayflick limit. For humans it is around 60 times. After that the important pieces of the DNA code start to be lost and the cell eventually cannot reproduce any more.
With cancer cells, the opposite occurs. For some reason cells begin to grow their telomeres so that their Hayflick limit is infinite -- they become immortal. Usually these cells are defective in other ways besides their telomeres and, by making more copies of this defect, they form a tumor. Because they outlive the normal cells, cancer is usually fatal.
In their attempts to make people live longer, scientists have learned how to save and restore telomeres with interventions such as medicine and stem cells. But, in doing so, they encounter another problem. Treating the whole person (i.e. collection of cells) to prevent telomere deterioration also promotes the immortality of pre-cancerous cells. These cells occur naturally and their defects usually cause their inability to divide and the cancer is thwarted. Bu if these cells are given immortality they will continue to grow and make copies of the defect.
A Two Prong Attack
In order o fight cancer and prolong life (since the two are entangled) we must find a way to preserve the telomere and at the same time prevent defective cells from proliferating. The potential solution came as a surprise.
Over the last three years, Gorbunova and Andrei Seluanov, research professor of biology at the University of Rochester, have worked to understand the similarities and differences of how varied but closely related species of rats deal with cancer. When Gorbunova and her team began investigating mole rat cells, they were surprised at how difficult it was to grow the cells in the lab for study.
The mole rat cells simply refused to replicate once a certain number of them occupied a space in the petri dish. Other cells, such as human cells, also cease replication when their populations become too dense, but the mole rat cells were reaching their limit much earlier than other animals' cells. Why?
Naked mole rats are strange, ugly, nearly hairless mouse-like creatures that live in underground communities. Unlike any other mammal, these communities consist of queens and workers more reminiscent of bees than rodents. Naked mole rats can live up to 30 years, which is exceptionally long for a small rodent. Despite large numbers of naked mole-rats under observation, there has never been a single recorded case of a mole rat contracting cancer. Adding to their mystery is the fact that mole rats appear to age very little until the very end of their lives.-- Gorbunova.Until Gorbunova and Seluanov's research, the prevailing wisdom had assumed that an animal that lived as long as we humans do needed to suppress the telomere activity to guard against cancer. Telomeres help cells reproduce, and cancer is essentially runaway cellular reproduction. An animal living for 70 years has a lot of chances for its cells to mutate into cancer.
In 2006, Gorbunova made the surprising discovery that telomerase -- the enzyme that makes up the telomeres -- is highly active in small rodents like the mole rat.
Discovering Nature's Trade-offs
Gorbunova points out that a rat's life expectancy is often shortened by other factors in nature, such as accidents or being hunted. Since they were not expected to live long enough to get cancer anyway, the regrowth of their telomeres for improved healing of wounds and injuries made sense. But the naked mole rats live longer than other rats -- sometimes 30 years. They should show signs of cancers and tumors, but they do not.
What is different about these naked mole rats? Do they contain some chemical that prevents cancer from starting?
"Since cancer is basically runaway cell replication, we realized that whatever was doing this was probably the same thing that prevented cancer from ever getting started in the mole rats."The special gene
--Gorbunova.
Gorbunova believes she found the primary reason these small animals are staying cancer-free, and it appears to be a kind of "overcrowding early-warning gene" that the naked mole rat expresses in its cells.
The findings, presented first in 2009 in The Proceedings of the National Academy of Sciences, showed that the mole rat's cells express a gene called p16 that makes the cells "claustrophobic," stopping the cells' proliferation when too many of them crowd together, cutting off runaway growth before it can start. This is why the cells refused to grow beyond a certain density in the petri dish.
It is a new way of thinking about cancer and the various strategies for eliminating it. Previous work has sought to kill the cancerous cells as soon as they begin to grow. This often involved toxins which are given to the patient to kill the cancer, but which have negative side-effects on healthy cells. The technique which shuts down reproduction when a certain tumor size is reached seems to be a more natural response as it does not involve toxins.
The effect of p16 is so pronounced that when researchers purposely mutated the cells to induce a tumor, the cells' growth barely changed, whereas regular mouse cells became fully cancerous.
But there is more to the picture.
A single molecule with a tongue-twisting name
Seluanov and Gorbunova discovered that these naked mole rats are protected from cancer because their tissues are very rich with high molecular weight hyaluronan (HMW-HA) [below].
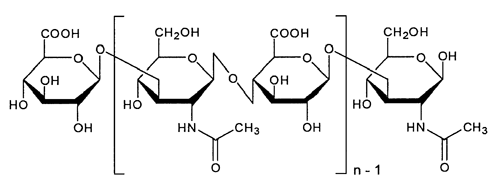
The biologists' focus on HMW-HA began after they noticed that a gooey substance in the naked mole rat culture was clogging the vacuum pumps and tubing. They also observed that, unlike the naked mole rat culture, other media containing cells from humans, mice, and guinea pigs were not viscous.
"We needed to understand what the goo was," said Seluanov.
Gorbunova and Seluanov identified the substance as HMW-HA, which caused them to test its possible role in naked mole rat's cancer resistance.
Seluanov and Gorbunova observed that when HMW-HA was removed, the cells became susceptible to tumors, confirming that the chemical did play a role in making naked mole rats cancer-proof.
Gene identified... Now what?
The Rochester team also identified the gene, named HAS2, responsible for making HMW-HA in the naked mole rat. Surprisingly, the naked mole rat gene was different from HAS2 in all other animals. In addition to its production, the naked mole rats were very slow at recycling HMW-HA, which contributed to the accumulation of the chemical in the animals' tissues.
The next step will be to test the effectiveness of HMW-HA in mice. If that test goes well, Seluanov and Gorbunova hope to try the chemical on human cells.
"There's indirect evidence that HMW-HA would work in people. It's used in anti-wrinkle injections and to relieve pain from arthritis in knee joints, without any adverse effects. Our hope is that it can also induce an anti-cancer response.A lot of cancer research focuses on animals that are prone to cancer. "We think it's possible to learn strategies for preventing tumors by studying animals that are cancer-proof."
--Seluanov
Future research from the Gorbunova and Seluanov labs will focus on determining whether the HMW-HA from naked mole rats may have clinical value for either treating or preventing cancer in humans.
Perhaps in the not so distant future the human genome will incorporate the naked mole rat's HAS2 gene, hopefully without the goo.

